VUE Biomarker Panels
Gain fast and reliable insights for your specific biological hypothesis using pre-optimized panels configured for you through our VUE portfolio, with:
- InSituPlex® technology enabling consistent assay performance with high sensitivity and specificity
- Ready-to-use kits
- Configurable panels with fast turnaround times
- Same-slide H&E staining
- Expert assistance with seamless onboarding of InSituPlex® assay workflows
- Our team of assay development specialists for custom biomarker and panel development
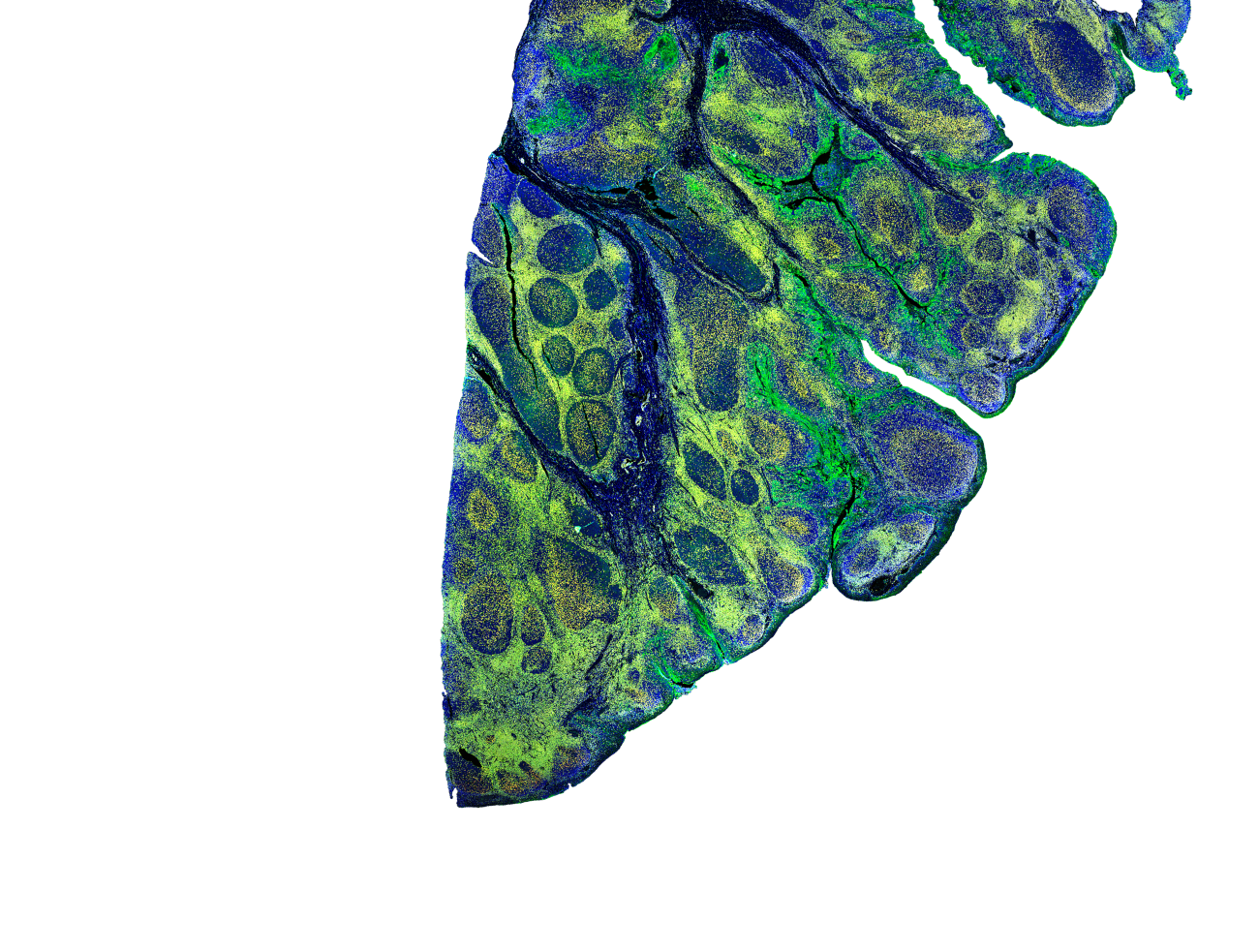
InSituPlex® Technology
Why Tissue Plex with InSituPlex?
Ultivue provides researchers and scientists with multiplex biomarker assays and advanced image informatics for tissue phenotyping and digital pathology. Our proprietary InSituPlex® technology enables advanced exploration and interrogation of tissue samples for precision medicine research. These highly customizable solutions and scientific consultative approach strengthen and accelerate biomarker discovery and drug development programs.
InSituPlex® technology requires only a single antigen retrieval step before applying a mixture of DNA barcoded antibodies, which then bind to the desired array of antigens. After amplification, fluorescent probes are attached to those barcodes, allowing for visualisation of the model using fluorescent microscopy. The InSituPlex technology is available in 4, 8, 12-plex panel options.

Don’t see your biomarker of interest? Reach out to us and we will develop the custom marker and panel for you.
Get the spatial insights that matter to you through customized biomarker panels.
Contact Sales
